Laboratory work typically involves constant adaptation to new experimental findings. While we may plan to pursue certain research ideas at the beginning of a project, our follow-up questions are nearly always dependent on the early set of experimental results, and they are subject to change. As such, scientific project management requires a workflow that can be flexible.
Agile is an iterative approach to project management that promotes velocity and adaptability. Principles from agile project management let scientific teams continuously improve their research workflows by encouraging laboratory members to quickly adapt to new initiatives, work on tasks in parallel, and continuously communicate with each other.
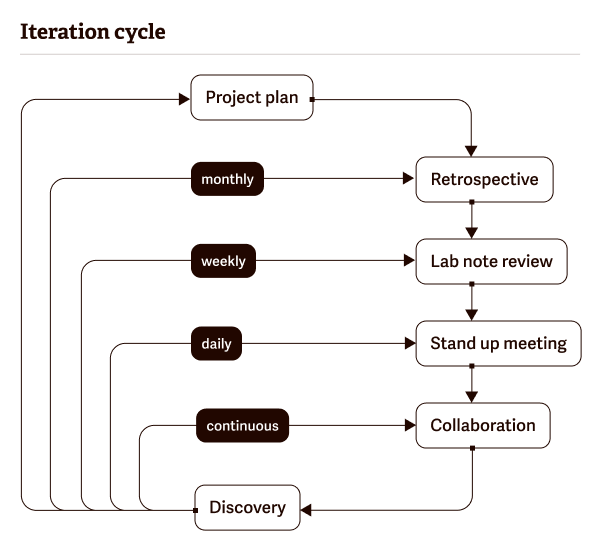
Basic principles of agile lab project management
There are six steps to agile lab project management:
- Project planning
- Product roadmap creation
- Release planning
- Sprint planning
- Daily stand-ups
- Sprint review and retrospective
Before diving into the details of each of these steps, there are a few agile-specific terms to become familiar with:
- Product: In commercial settings, “product” typically means a product for the market. However, in a scientific setting, a product could be a specific experimental result, a completed manuscript, a new drug, etc.
- Backlog: A list of tasks to be prioritized.
- Sprint: A short period (2-6 weeks) when a team completes a specific set of tasks from the backlog.
- Sprint review: A meeting during which the group assesses the original planned and executed tasks during a specific sprint.
- Retrospective: A meeting during which the group discusses what went well and what could have gone better during a specific sprint.
Altogether, the agile process takes a group from initial planning to completion, along with a critical analysis of how project steps could be improved in the future.
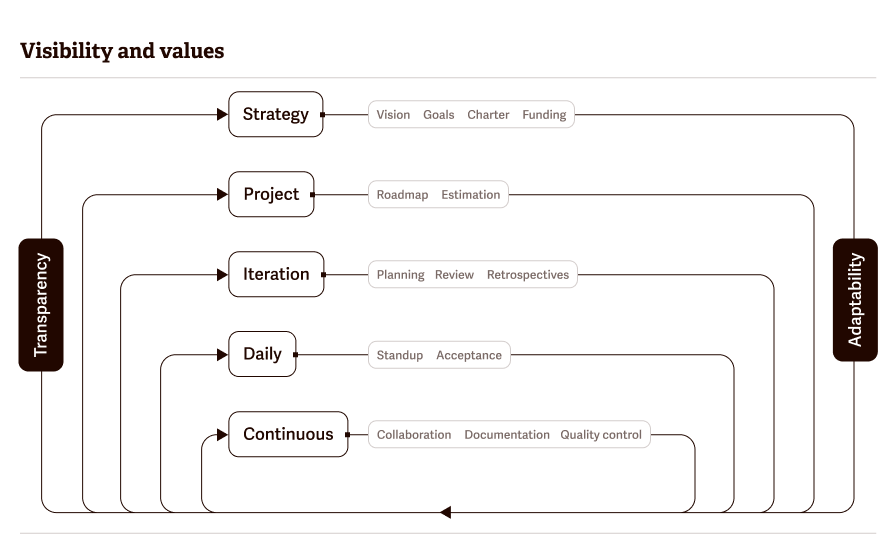
Each workflow step can be broken down into different parts, and laboratories can adapt these steps to their projects.
Setting the stage for agile research: Practical example
Below, we provide a real-world example for each stage of the agile research project management process. The example illustrates a laboratory group developing a patient-derived xenograft model of pancreatic cancer in mice.
1: Project planning
During the project planning stage, team members work together to define and assess the project’s overall goals and why the specific project is important. For example, if a group initiates a project for optimizing a chemical synthesis protocol, the impact may be that the process would make future syntheses cheaper and faster. Project planning helps scientists better understand the big picture of the research project.
Setting a clear direction is critical for doing science as a team. Defining these goals and objectives early in the project development process provides each member with a clear view of the project’s end goals. This empowers individuals to make daily decisions, helps teams prioritize tasks, and motivates all group members to work together toward a shared purpose.
Teams should discuss the following topics:
- Determine methods for note, protocol, and data storage
- Establish mechanisms for open communication between members
The team comes together to formulate research plans during the project planning phase. They establish that they want to develop a patient-derived xenograft model of pancreatic cancer in mice. They define this project as significant because the model will allow the group to better characterize physiological responses to a new drug. The group establishes that they will need a variety of expertise on the team, including a clinician to obtain tumor samples, a group of scientists to work on cell culture and mouse work, a veterinarian to help care for the mice, and scientists for genetic and histopathological analyses of tumor tissues. The group also decides to use Colabra to share all data relevant to the project.
2: Product roadmap creation
The product roadmap creation stage shapes the direction of the project. The roadmap forces research leaders to break down a larger goal into specific key milestones, articulate a series of experiments necessary to obtain specific results, and allocate resources accordingly.
The main goal of project roadmap creation is to assess how long certain objectives may take and to break down the project into smaller tasks that can be accomplished in shorter periods. In addition, the group will need to assess dependencies between the tasks to allocate resources optimally toward any perceived bottlenecks. Groups will need to determine which tasks need to be performed sequentially and if any can be performed in parallel.
There are several decisions that need to be made:
- Initial study hypothesis;
- Specific sets of experiments and tasks that need to be performed;
- Specific analyses that are needed;
- Designated people with the expertise to perform the required experiments and analyses;
- Confirm all members understand their roles; and
- Establish a general timeline for milestone completion.
The team starts to plan its roadmap, and it decides that there are two main milestones and several specific tasks. Milestone 1: Obtain tumor samples and generate patient-derived xenograft cell lines. Task 1.1: Contact clinician to obtain tissue samples. Task 1.2: Coordinate transfer of tissues. Task 1.3: Develop protocols for handling tissues and extracting cells. Task 1.4: Develop protocols for cell culture, freezing, and thawing. Task 1.5: Obtain tissues. Milestone 2: Implant cells in mice and monitor tumors. Task 2.1: Prepare cells and inoculate mice. Task 2.2: Monitor tumor growth. Task 2.3: Collect tumor tissues. Task 2.4: Perform histopathological analysis. Task 2.5: Perform flow cytometric analysis. Task 2.6: Perform genetic analysis.
3: Release planning
The release planning stage defines the necessary completion of specific milestones before releasing the product. In the scientific setting, these milestones can vary – for example, if the end goal is to submit a manuscript, then the major milestone of that project may include finalizing the set of experimental data. If a group aims to initiate a clinical trial for a new drug, a major milestone would be to complete an Investigational New Drug form with the FDA.
An important aspect to remember in planning in science is that experiments may not always work the way the team thought they would. Groups need to be realistic in estimating their timelines and milestones.
Teams should discuss the following topics:
- Define end products (or anticipated results)
- Discuss backup plans
The group establishes the release conditions for their project: 1. Successfully establish viable patient-derived xenograft pancreatic cell lines. 2. Successfully develop tumors in mice from the cell line. 3. Complete genetic and histopathological analysis on collected tumors. The group also brainstorms backup plans if some of the experimental tasks are met with challenges as they get started.
4: Sprint planning
In sprint planning, the group identifies the tasks they will work on first and determines a timeline for sprint completion. Sprints can occur in as little as 1-2 weeks, or as long as 6 weeks.
In the sprint planning stage, the group assesses and prioritizes the objectives and tasks created during the roadmap creation stage. During roadmap creation, dependencies between the tasks should have also been defined, which will help determine the order and priority of each task.
Once sprint planning is completed, the first sprint can begin.
The group uses their goals created during the roadmap creation stage to start planning their sprint. Since their second goal depends on completing the first goal, they focus on completing tasks within goal one first. The group establishes that during their 6-week sprint, they will first contact a clinician to obtain tissue samples (Task 1.1) and then work on Tasks 1.2, 1.3, and 1.4 in parallel. They recognize that there may be some hurdles in getting Tasks 1.1 completed, such as regulatory approvals, clinician timelines, and shipment considerations. They make a note to revisit their timeline in 2 weeks to adapt future tasks as needed.
5: Daily stand-ups
Stand-ups are intended to be short meetings to address the tasks that are being completed during the sprint. As experiments and other laboratory tasks may take a few days to complete, a research group may decide to hold weekly stand-ups instead. These meetings are not intended to be exhaustive but are rather intended to provide the group with a brief recap of (1) what was recently completed, (2) what is being completed next, (3) and potential problems that have arisen.
Stand-ups are important because they allow projects to leverage the entire team’s full cognitive ability and experience. In addition, these meetings create a culture of transparency and accountability between members, which builds trust within the group. Another important aspect of stand-ups is that they enable members to get feedback quickly, which helps members overcome hurdles to complete their tasks. It is important that laboratory leaders create an environment that encourages members to ask for feedback and help, not just report a status.
In evaluating the tasks involved in developing the mouse model, the group decides to hold weekly stand-ups. They choose a dedicated time, Tuesdays at 9 AM. During their first stand-up, one member reports that they successfully scheduled the meeting with the clinician (Task 1.1). The team marks the task as complete and moves to Tasks 1.2, 1.3, and 1.4.
6: Sprint review and retrospective
After the sprint is complete, the sprint review assesses the results and outcomes of the sprint, and the retrospective assesses potential workflow and communication improvements.
In a sprint review, the group shares results from the experiments, potential issues with methodologies, and strategies for overcoming these issues in the future. During this review, the group may identify additional tasks for future sprints.
In a retrospective, the group focuses on personnel and workflow improvements. For example, a research group may have been communicating experimental results via email, and these results were getting “lost” in members’ mailboxes. Thus, the group discusses new communications strategies, such as a document sharing program, to share information during the next sprint. The team determines that a more open and accessible data sharing method is better for the lab’s long-term goals, as these methods would reduce the likelihood of institutional data loss.
The sprint review and the retrospective are critical for experimental groups to continually optimize future sprints.
The group has completed their first sprint, and they are now ready to hold their sprint review and retrospective. During the sprint review, one member provides an update that they have identified a clinician to send them tumor samples. The clinician is excited to start a collaboration with the group. However, one hurdle that arose was that the group did not have the appropriate ethical clearance to obtain the tissue samples. The group determines they need to add a new task; “Update Institutional Review Board documentation for obtaining tumor samples.” During their retrospective, the group decides that weekly stand-ups are too frequent for the beginning stages of this project. They decide to move forward with stand-ups every two weeks. As the project picks up pace, the team may make meetings more frequent again.